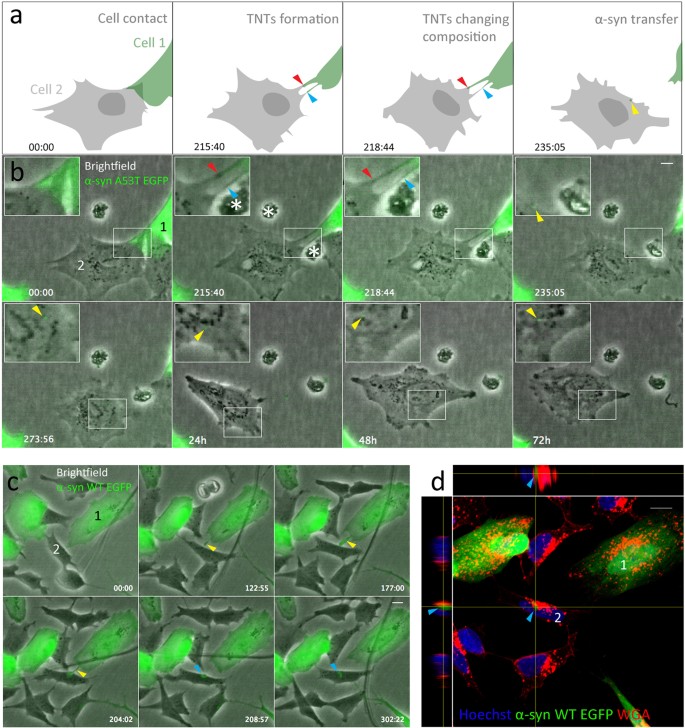
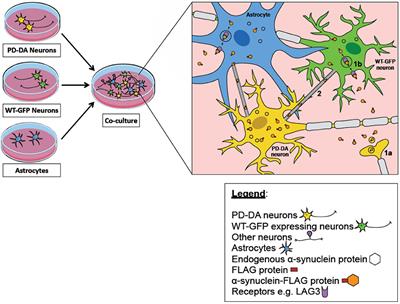
α-synuclein transfer through tunneling nanotubes occurs in SH-SY5Y cells and primary brain pericytes from Parkinson's disease patients | Scientific Reports
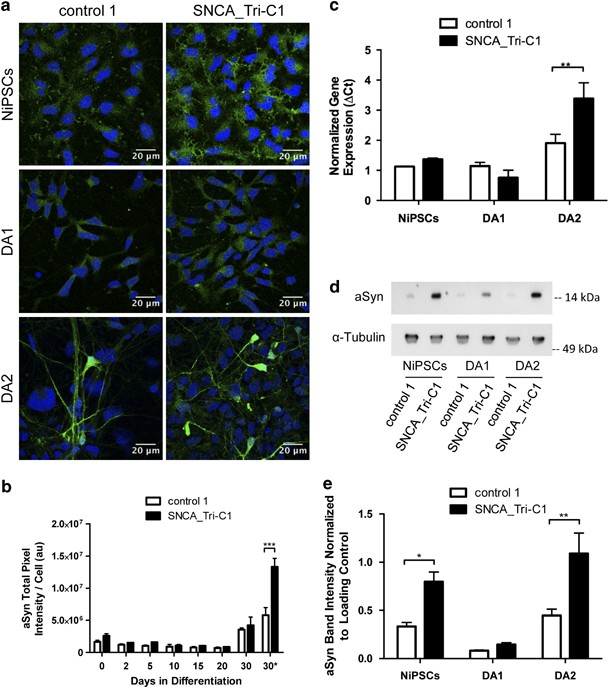
Frontiers | Patient-Derived Induced Pluripotent Stem Cells and Organoids for Modeling Alpha Synuclein Propagation in Parkinson's Disease | Cellular Neuroscience
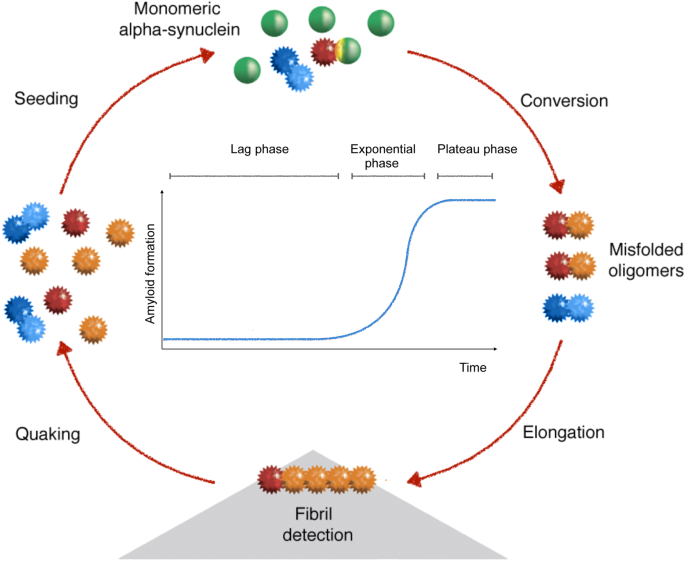
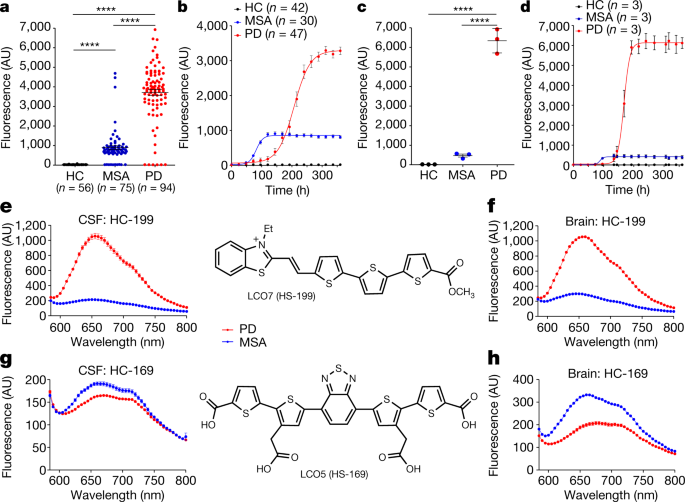
Effect of the micro-environment on α-synuclein conversion and implication in seeded conversion assays | Translational Neurodegeneration | Full Text

Early synaptic dysfunction induced by α-synuclein in a rat model of Parkinson's disease | Scientific Reports
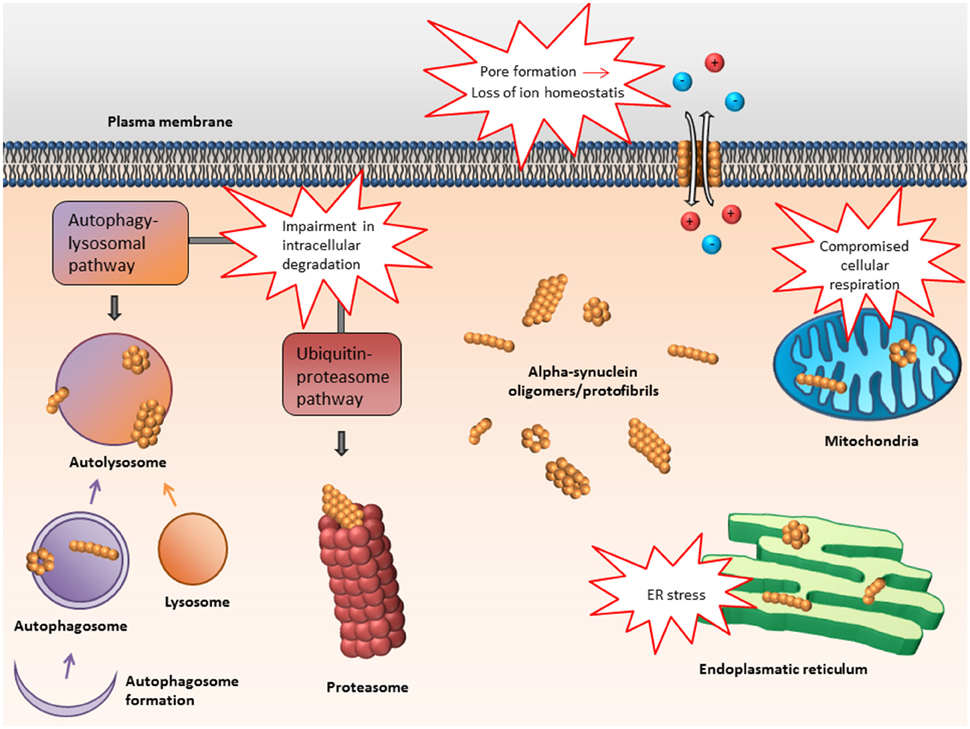
Frontiers | Alpha-Synuclein Oligomers—Neurotoxic Molecules in Parkinson's Disease and Other Lewy Body Disorders | Neuroscience

Subcellular localization of alpha-synuclein aggregates and their interaction with membranes Miraglia F, Ricci A, Rota L, Colla E - Neural Regen Res

A role for α-Synuclein in axon growth and its implications in corticostriatal glutamatergic plasticity in Parkinson's disease | Molecular Neurodegeneration | Full Text

α-Synuclein oligomers mediate the aberrant form of spike-induced calcium release from IP 3 receptor | Scientific Reports

Subcellular localization of alpha-synuclein aggregates and their interaction with membranes Miraglia F, Ricci A, Rota L, Colla E - Neural Regen Res
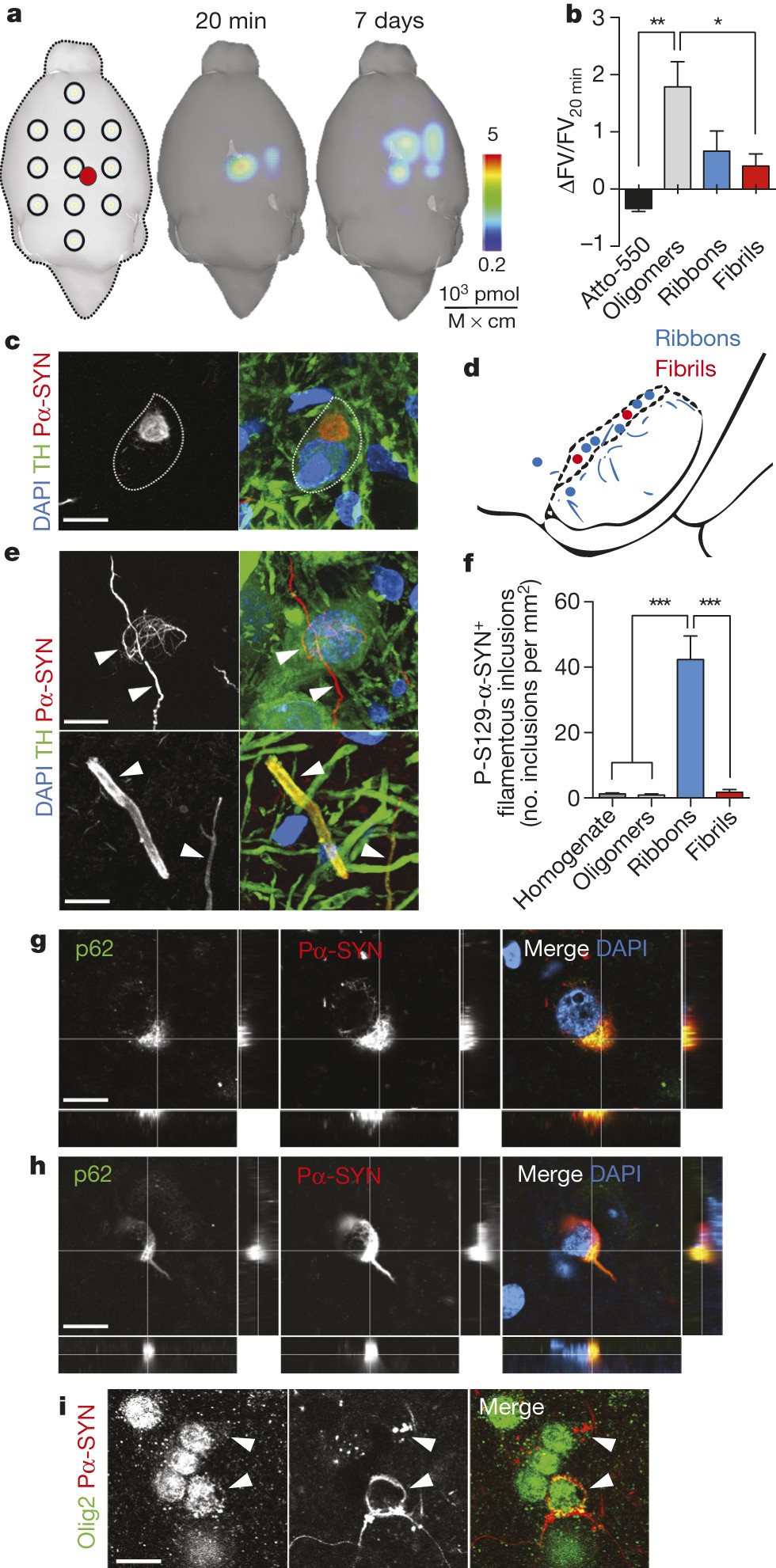
α-Synuclein strains cause distinct synucleinopathies after local and systemic administration | Nature
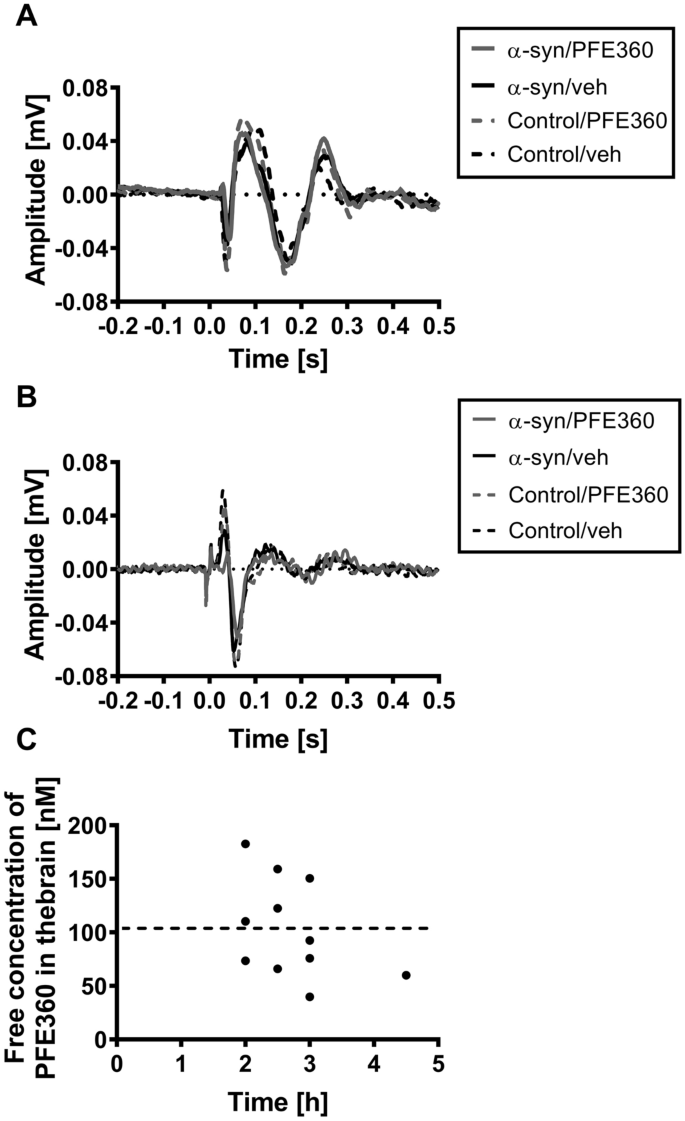
Classification of α-synuclein-induced changes in the AAV α-synuclein rat model of Parkinson's disease using electrophysiological measurements of visual processing | Scientific Reports
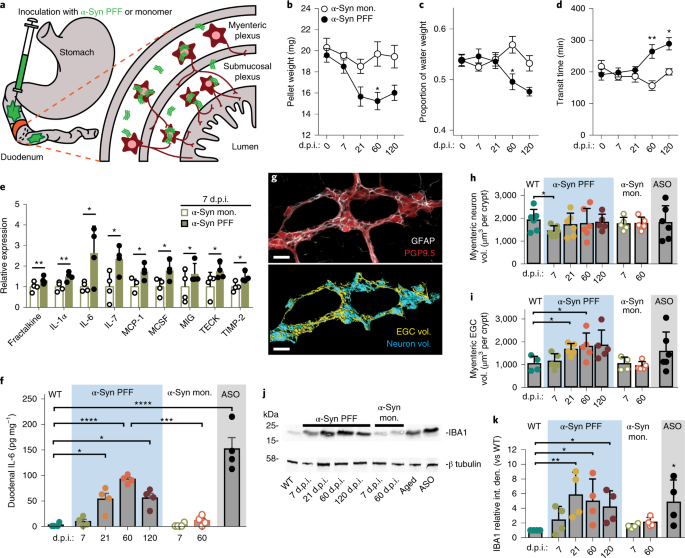
Gut-seeded α-synuclein fibrils promote gut dysfunction and brain pathology specifically in aged mice | Nature Neuroscience
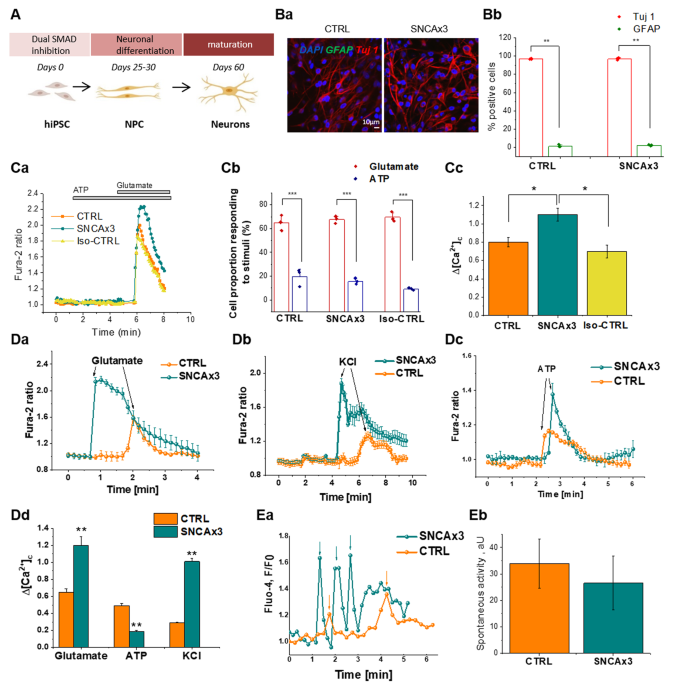
Alpha synuclein aggregation drives ferroptosis: an interplay of iron, calcium and lipid peroxidation | Cell Death & Differentiation

Frontiers | α-Synuclein: A Multifunctional Player in Exocytosis, Endocytosis, and Vesicle Recycling | Neuroscience
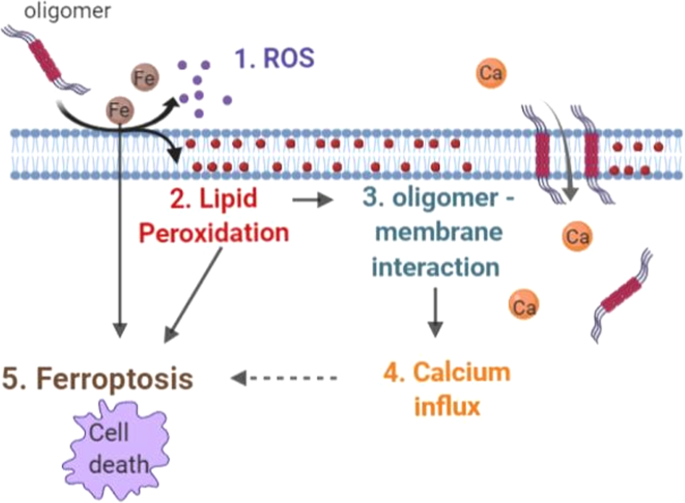
Alpha synuclein aggregation drives ferroptosis: an interplay of iron, calcium and lipid peroxidation | Cell Death & Differentiation
Single-Channel Electrophysiology Reveals a Distinct and Uniform Pore Complex Formed by α-Synuclein Oligomers in Lipid Membranes
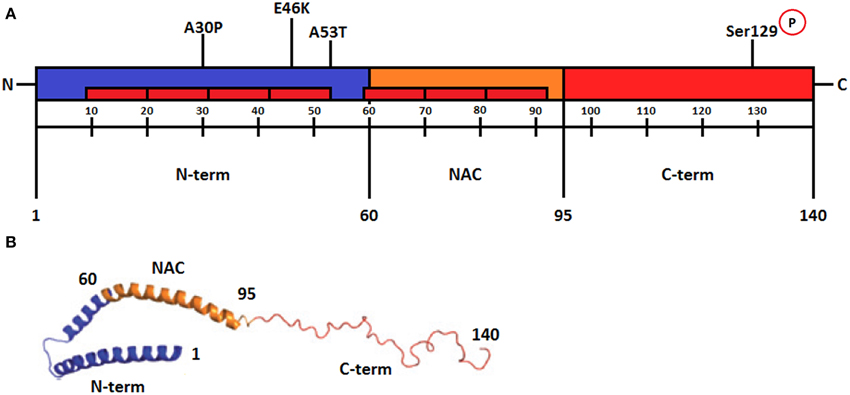
Frontiers | Features of alpha-synuclein that could explain the progression and irreversibility of Parkinson's disease | Neuroscience

Seeding and transgenic overexpression of alpha‐synuclein triggers dendritic spine pathology in the neocortex | EMBO Molecular Medicine
Single-Channel Electrophysiology Reveals a Distinct and Uniform Pore Complex Formed by α-Synuclein Oligomers in Lipid Membranes
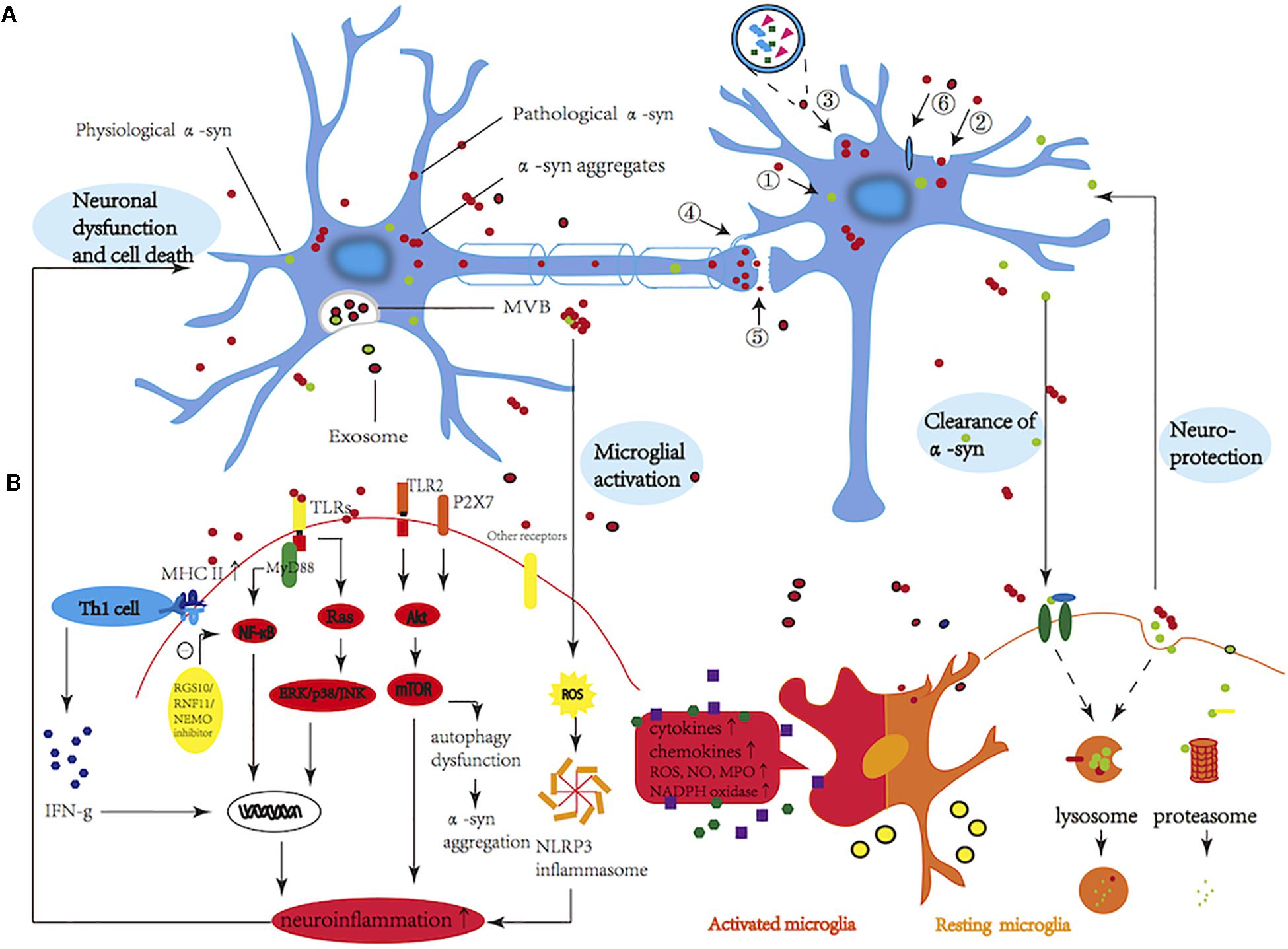
Frontiers | New Perspectives on Roles of Alpha-Synuclein in Parkinson's Disease | Frontiers in Aging Neuroscience
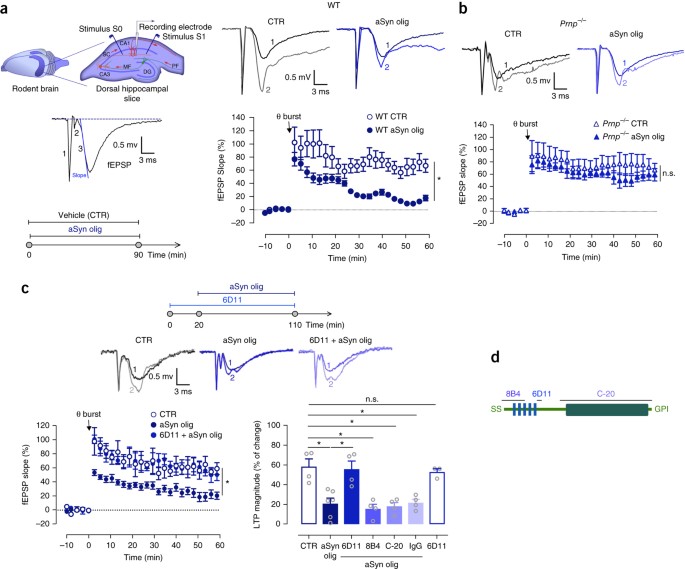
α-synuclein interacts with PrP C to induce cognitive impairment through mGluR5 and NMDAR2B | Nature Neuroscience